Annual American Society of Clinical Oncology meeting held virtually in June
The American Society of Clinical Oncology (ASCO) conference is one of the largest and most diverse meetings of clinical oncologists held each year. This year’s meeting, which took place June 4-8, was held fully online and included more than 2,500 presentations. Guided by the theme, “Equity: Every Patient. Every Day. Everywhere,” the conference was expected to draw more than 44,000 attendees from more than 130 countries.

Research by Kaiser Permanente investigators from the Division of Research (DOR) and physicians from The Permanente Medical Group (TPMG) was represented throughout the program. In addition, on June 8, The ASCO Post covered research based on the ongoing Kaiser Permanente Northern California Pathways Study of breast cancer survivors that found a relationship between vitamin D levels at the time of diagnosis and patient outcomes. The ASCO Post story included a quote from DOR research scientist Lawrence Kushi, ScD, the study’s senior author, who noted, “These findings highlight not just the role of vitamin D in breast cancer prognosis, but also the contribution of and need for prospective studies in cancer survivors to complement clinical trials.” DOR’s Marilyn Kwan, PhD, was a co-author of the study.
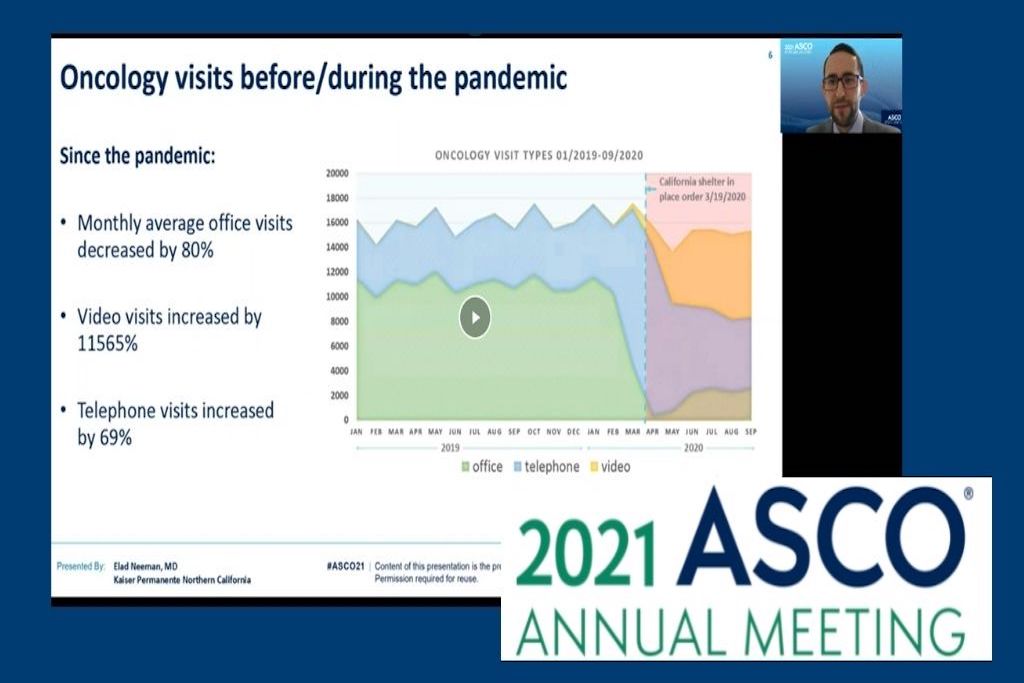
Elad Neeman, MD, a second-year TPMG oncology fellow, received an Annual Meeting Merit Award, and his oral presentation, “The future of tele-oncology: Trends and disparities in telehealth and secure message utilization in the COVID-19 era,” was among the top 9 studies selected by ASCO’s Care Delivery and Regulatory Policy section. Co-authors included Liisa Lyon, MS, Carol Conell, PhD, and Mary Reed, DrPH, of the DOR; and Hongxin Sun, MS, Deepika Kumar, MD, Tatjana Kolevska, MD, Dinesh Kotak, MD, Tilak Kumar Sundaresan, MD, and Raymond Liu, MD, of TPMG.
TPMG’s Raleigh Fatoki, MD, MBA, a second-year resident, was 1 of 7 medical residents from underrepresented populations in medicine who received an Annual Meeting Research Award. His study, Characterization of smoldering multiple myeloma: a population-based cohort study, was included as an abstract.
Significant studies also included:

— NEJM co-authorship by TPMG’s Jennifer Marie Suga, MD, of results from the OlympiA study, which is likely to change the standard of care for breast cancer with BRCA mutations and expand genetic testing to more breast cancer patients.
— Minggui Pan, MD, PhD, an adjunct investigator with the DOR and a TPMG oncologist/hematologist, and DOR research scientist Laurel Habel, PhD, were part of a team that included TPMG physicians that presented two posters representing studies from the DOR Strata genomic researchers and the TPMG genomic oncology committee that suggest different TP53 gain of function mutations are associated with very different clinical outcome and that TP53 mutations are associated with decreased prevalence of Ras, PI3KCA mutation and MSI-high in patients with metastatic colorectal cancer.
— DOR research scientist Bette Caan, DrPH, co-authored a paper in the Developmental Therapeutics track that looked at whether oxaliplatin pharmacokinetics explains associations between body composition and chemotherapy toxicity risk in older adults with gastrointestinal cancers.

— A Kaiser Community Benefit Grant funded research by TPMG and DOR — led by TPMG’s Elizabeth Shurell Linehan, MD, MPhil — that was included in the Disparities/Access to Care track examining racial disparities in male breast cancer.
Seven large, national studies presented at ASCO also included TPMG physicians as co-authors, highlighting the work of the Kaiser Permanente Oncology Clinical Trials Program. “We would like to thank all of the patients with cancer who enrolled in these important clinical trials,” said Suga. “Their participation is key to moving the cancer research field forward.”
— Melanoma: SWOG 1404: Final analysis of overall survival and relapse-free-survival in the intergroup S1404 phase III randomized trial comparing either high-dose interferon or ipilimumab to pembrolizumab in patients with high-risk resected melanoma.
— Prostate: SWOG S1216: A phase III randomized trial comparing androgen deprivation therapy (ADT) plus TAK-700 with ADT plus bicalutamide in patients with newly diagnosed metastatic hormone-sensitive prostate cancer.
— Breast: NSABP B-42: Breast Cancer Index and prediction of benefit from extended aromatase inhibitor therapy in HR+ breast cancer
— Breast: NSABP B-42: Utility of the 70-gene MammaPrint assay for prediction of benefit from extended letrozole therapy.
— Breast: SWOG 1200: KPOCT enrolled 56 of the 226 patients enrolled in the study, presented as a poster, which found that acupuncture reduced joint symptom pain related to aromatase inhibitors.
— Colorectal: EAQ162CD: Baseline and short-term financial burden in colorectal cancer treated with curative intent.
— GI: NSABP C-08: Examination of the tumor immune microenvironment with multispectral immunofluorescence: Association of markers with prognosis and bevacizumab benefit.
— Lung: SUMMIT: Neratinib efficacy in a subgroup of patients with EGFR exon 18–mutant non-small cell lung cancer and central nervous system involvement: Findings from the SUMMIT basket trial.
— Ovarian: AVB-500: Phase 1b study of GAS6/AXL inhibitor (AVB-500) in recurrent, platinum-resistant ovarian carcinoma.
Additional presentations by Kaiser Permanente fellows and residents:
— Adnan Kahn, MD: Clinical presentation of multiple myeloma by race/ethnicity and Asian subgroup in an integrated healthcare system.
— Andrew Lynch, MD: Characteristics of lymphoma presenting initially as a neck mass referral to otolaryngology.
— Tiffany Seto, MD: Treatment patterns for patients with metastatic or recurrent head and neck cancer in a large integrated health-care system.




Comments (0)